
The Long War Against the Iron Pirates
Disease is an act of piracy. When microbes infect us, they steal our resources so they can thrive at our expense. We fight them off with direct attacks, using an army of immune cells and antibodies. But we also have subtler countermeasures: we can, for example, deprive them of the nutrients they need.
Iron is one such nutrient. Many of the most important enzymes only work when they embrace iron atoms, and that’s true for blue whales and bacteria alike. So, when bacteria infect us, they try to scavenge iron from our bodies. We, in turn, try to halt their advances by withholding this nutrient.
Matthew Barber and Nels Elde from the University of Utah School of Medicine have found clear signs of this war over iron in two molecules: transferrin, an animal protein that stores iron in a tight embrace, and TbpA, a bacterial protein that literally snatches iron from transferrin’s grasp.
These proteins have gone through repeated bursts of rapid evolution. Time and again, bacterial TbpAs have evolved to better prise iron from our grasp, while the transferrins of humans and other apes have evolved to clutch the element more tightly. We have been waging this war against iron pirates for millions of years, and neither side shows any side of giving ground.
Elde was interested in evolutionary arms races between infectious microbes and the hosts that they trouble. These conflicts leave their mark on the the genes that drive our immune systems. One such gene, for example, might recognise a telltale molecule on the surface of a virus. The virus adapts by changing those surface molecules to avoid detection, and the gene counter-adapts to spot the virus’s new guise. On and on they go, thrusting and parrying through the generations.
These genes tend to evolve in rapid and repeated bursts—and that’s exactly what Barber and Elde saw when they compared the transferrins of 21 species of primates. “Transferrin was acting as if it was an immunity gene,” says Elde. It was adapting quickly and repeatedly to ever-changing adversaries.
Transferrin acts like a thermostat for iron. It stores the element, keeping it in stock while also stopping toxic levels from building up in our bodies. It looks like a peanut, with two lobes (N and C) that each grasps a single iron ion. Their hold is so delicate that you’d expect the lobes to change very little over time. The N-lobe meets that expectation. It looks the same, “no matter what primate you pull out of the hat,” says Elde.
The C-lobe is different. It has been a hotbed of change, and especially among apes. Barber and Elde found 18 parts of transferrin that have rapidly evolved in primates, and 16 of these are in the C-lobe. On their own, these changes have no obvious purpose. But their significance became clear when the team visualised transferrin next to the TbpA protein from Neisseria meningitidis—a microbe that causes bacterial meningitis.
TbpA is shaped like a claw. It seizes transferrin, prises it apart, and relieves it of its iron. This only works because the shape of the claw conforms exactly to the shape of its transferrin target. The molecules touch each other at several specific points and, sure enough, these are exactly the same parts of transferrin that evolve most rapidly. The protein changes shape to avoid getting snatched.
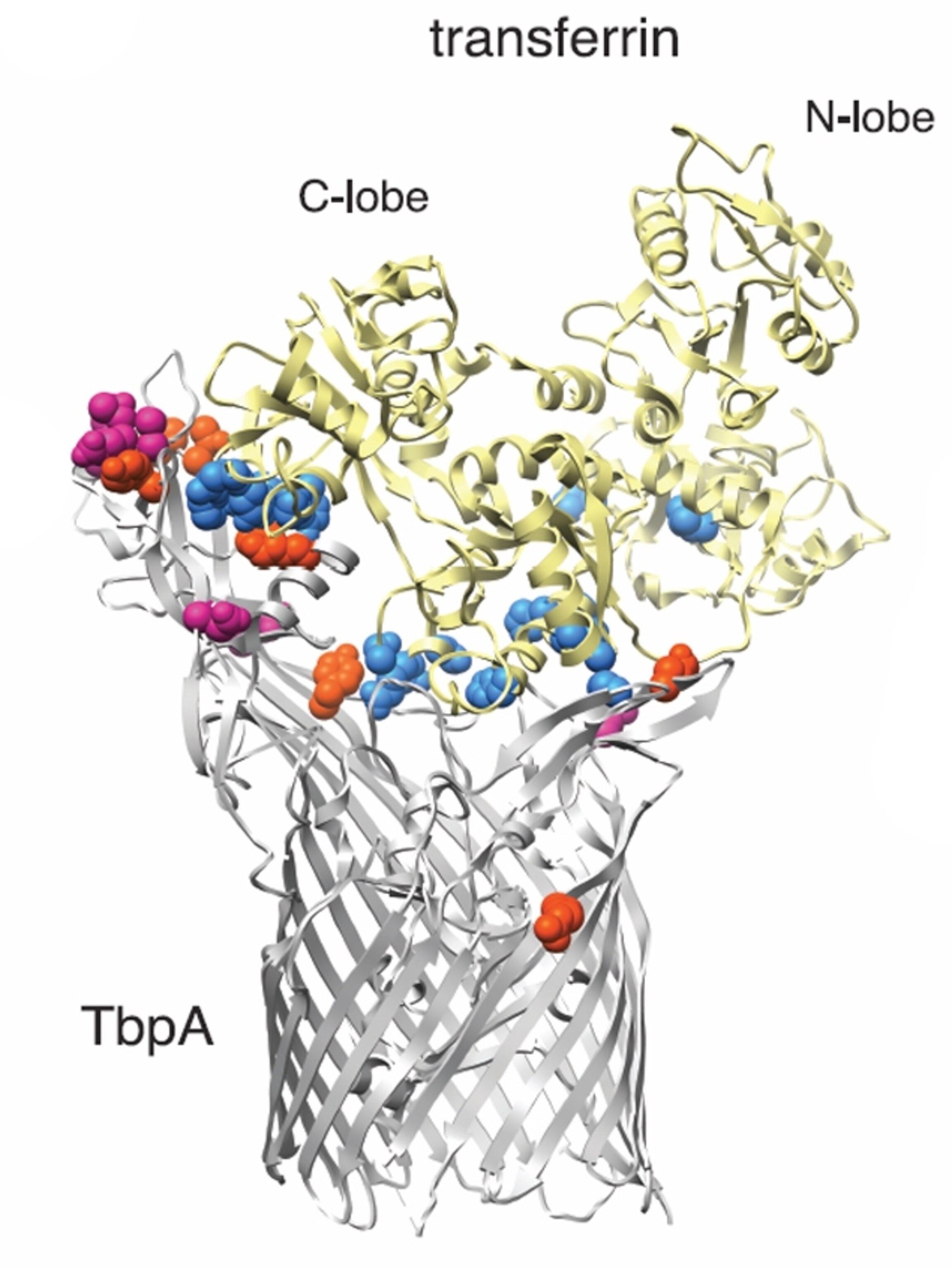
But TbpA fights back. Barber and Elde compared TbpA genes in several strains of N.gonorrhoeae, which causes gonorrhoea, and Haemophilius influenzae, which causes flu-like symptoms. These bacterial proteins showed the same signs of recurring rapid evolution, especially in the parts that recognise and grab transferrin. Their ‘prey’ changes shape to slip through their grasp, but they adapt by changing their hold.
This evolutionary arms race has played out over 40 million years at least. Barber and Elde demonstrated this by the purifying transferrins from humans, gorillas, chimps, orang-utans, gibbons and baboons, and pitting them against TbpAs from N.gonorrhoeae and H.influenzae. These bacterial proteins recognised transferrins from humans and gorillas, but not the other species. This means that their owners can steal iron from human cells, but not from chimp cells—a striking difference, considering that chimps are our closest relatives.
“Once we saw the difference with chimps, we thought: We should really look at human diversity,” says Elde. “And we didn’t have to look far.” It turned out that not all transferrins are created equal. Humans have two major types: the C1 version, which is the most common, and the C2 version, which is found in 6 to 26 percent of people. Scientists have known about these two versions for a long time, but no one really knew why they co-existed. They differ by a single DNA letter and they certainly both bind iron with equal vigour.
Once again, this mystery became clearer once Barber and Elde pitted the transferrins against iron-stealing microbes. They found that N.meningitidis and N.gonorrhoeae recognise both the C1 and C2 transferrins, but H.influenzae struggles to capture the C2 version. So the number of people who carry the C1 or C2 versions will depend on how common and how deadly these various microbes are.
All of these results suggest that transferrin is part of a nutritional immune system—a set of molecules that prevent disease by starving microbes of essential nutrients. This is an old concept. The scientists who first purified transferrin in 1946 hinted at it. But Barber and Elde have shown that nutritional immunity is important enough to have left a noticeable evolutionary imprint in our genomes.
It might be possible to use this knowledge for our benefit. At a time when bacteria are becoming increasingly resistant to antibiotics, scientists are looking for alternative ways of killing them. “People have been toying with providing patients with more transferrin,” says Elde. “There might be something to that, but if you’re using transferrin therapy, why not try the C2 version, or the one that arose in chimps, depending on which bug you’re trying to counterarct. We could reach into the genetic medicine cabinet and use solutions that we’ve naturally come up with through millions of years of evolution.”
And I, as per usual, am interested in whether the trillions in bacteria in our guts—the ones that are supposedly beneficial partners—are also iron pirates. Some of these mutualists have TbpA proteins too. Do they try to steal iron from our cells? Do we have ways of stopping them, to ensure a stable relationship? “When do these cross over from beneficial relationships into something more nefarious?” says Elde. “There could be this dynamic undercurrent where one day our friends become our enemies, and vice versa.”
Reference: Barber & Elde. 2014. Escape from bacterial iron piracy through rapid evolution of transferrin. Science http://dx.doi.org/10.1126/science.1259329
Go Further
Animals
- What La Palma's 'lava tubes' tell us about life on other planetsWhat La Palma's 'lava tubes' tell us about life on other planets
- This fungus turns cicadas into zombies who procreate—then dieThis fungus turns cicadas into zombies who procreate—then die
- How can we protect grizzlies from their biggest threat—trains?How can we protect grizzlies from their biggest threat—trains?
- This ‘saber-toothed’ salmon wasn’t quite what we thoughtThis ‘saber-toothed’ salmon wasn’t quite what we thought
- Why this rhino-zebra friendship makes perfect senseWhy this rhino-zebra friendship makes perfect sense
Environment
- What La Palma's 'lava tubes' tell us about life on other planetsWhat La Palma's 'lava tubes' tell us about life on other planets
- How fungi form ‘fairy rings’ and inspire superstitionsHow fungi form ‘fairy rings’ and inspire superstitions
- Your favorite foods may not taste the same in the future. Here's why.Your favorite foods may not taste the same in the future. Here's why.
- Are the Great Lakes the key to solving America’s emissions conundrum?Are the Great Lakes the key to solving America’s emissions conundrum?
- The world’s historic sites face climate change. Can Petra lead the way?The world’s historic sites face climate change. Can Petra lead the way?
History & Culture
- Hawaii's Lei Day is about so much more than flowersHawaii's Lei Day is about so much more than flowers
- When treasure hunters find artifacts, who gets to keep them?When treasure hunters find artifacts, who gets to keep them?
- Meet the original members of the tortured poets departmentMeet the original members of the tortured poets department
- When America's first ladies brought séances to the White HouseWhen America's first ladies brought séances to the White House
Science
- Should you be concerned about bird flu in your milk?Should you be concerned about bird flu in your milk?
- Here's how astronomers found one of the rarest phenomenons in spaceHere's how astronomers found one of the rarest phenomenons in space
Travel
- Are Italy's 'problem bears' a danger to travellers?Are Italy's 'problem bears' a danger to travellers?
- How to navigate Nantes’ arts and culture scene
- Paid Content
How to navigate Nantes’ arts and culture scene - This striking city is home to some of Spain's most stylish hotelsThis striking city is home to some of Spain's most stylish hotels
- Photo story: a water-borne adventure into fragile AntarcticaPhoto story: a water-borne adventure into fragile Antarctica