
The Fungus Behind the Frog Apocalypse Hides in Crayfish

Imagine that a new disease started wiping out mammals in their hundreds. Whether human or cow, bat or anteater, every species of mammal seems to succumb to this unfamiliar blight. No one understands how one infection could kill so many distantly related animals, but that doesn’t stop the bodies from piling up. Within decades, entire species go extinct. This doomsday scenario is only a partial fiction. It’s actually happening – the only difference is that it’s happening to amphibians and not mammals.
Since at least the 1990s, a fungus called Batrachochytrium dendrobatidisa fungus called Batrachochytrium dendrobatidis, or ‘Bd’ for short, has been slaughtering its way through the world’s frogs. It has severely slashed the populations of some 200 species, and dozens of these may already be completely wiped out. (Here’s a roll call of the dead.)
The disease it causes—chytridiomycosis—is like no other, in both the number of species it infects—some 600 amphibians so far and counting—and its ability to drive them to extinction. “Disease ecologists have been pulling their hair out trying to understand how a pathogen that dives its host to extinction could persist in the wild to infect the next host that comes by,” says Vance Vredenburg from San Francisco State University.
One possibility is that the fungus isn’t just the amphibian specialist that most people see it as. Maybe it uses other animals as reservoirs, where it can hide between amphibian infections. That would allow it to drive its usual hosts extinct, without itself fading into oblivion.
What could this mysterious reservoir animal be? There have been plenty of false leads, but no solid evidence that Bd infects anything other than amphibians. That is, until now. Through a combination of lab and field experiments, Taegan McMahon from the University of South Florida has found that the fungus can infect freshwater crayfish, which can then pass their infections on to amphibians.
“When I first heard about this, I was very sceptical,” says Vrendenberg. “I’ve been working on this since 2000, and I even get people telling me they’ve been infected by the chytrid fungus. But now I’ve read through the paper, they have really covered their bases.”

Vredenberg’s scepticism is understandable. One group supposedly found the fungus growing on Australian shrimp, but later retracted their claims. Others have found Bd on algae, ducks, and nematode worms, and shown that it can grow on feathers, bird feet, and reptile skin. But since the fungus kills amphibians by massively overwhelming them, the presence of a few spores says very little. None of these studies showed that the fungus actually infects these alternative animals, or spreads from them to amphibians.
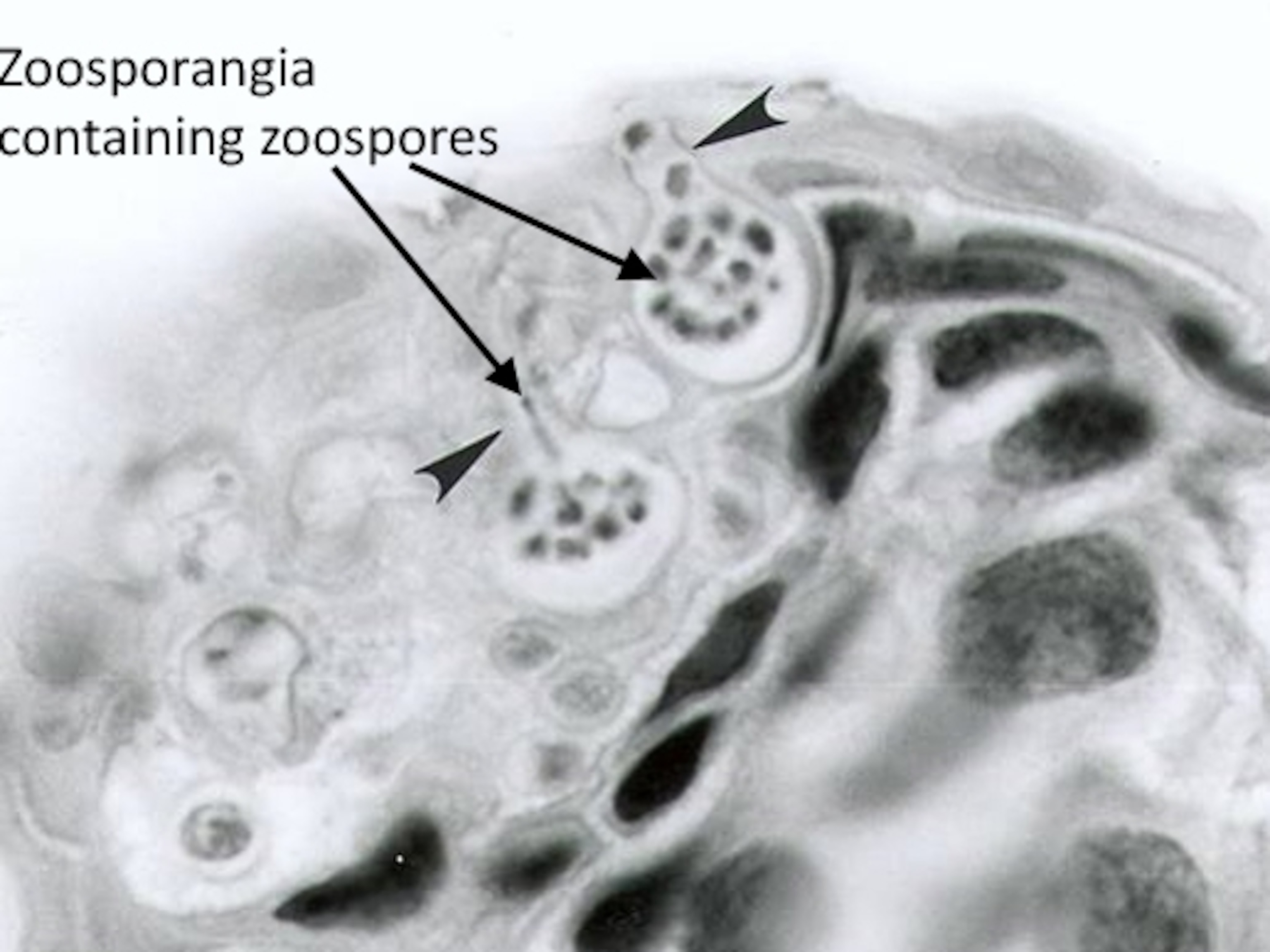
McMahon was far more thorough. She surveyed four species of crayfish in Louisiana and Colorado streams and found Bd zoosporangia—cases full of mobile spores—in 10 to 20 percent of them. These cases weren’t just sitting on the crayfish’s shells, but were deeply embedded inside their guts. They grew just as they do in frog skin, and McMahon confirmed through lab experiments that the fungus can infect the crayfish. Their guts become riotous masses of zoosporangia, and around a third of them die as a result.
In one of these surveys, done in September, McMahon found that one in every six crayfish was infected with the fungus, even though none of the local frogs were. This fits with previous studies showing that Bd kills amphibians in the spring, but is absent during the autumn. Maybe it hides in crayfish during this off-peak season, and jumps into frogs later. Certainly, the infected crayfish can harbour the fungus for months.
But McMahon hadn’t yet shown that the fungus could jump from crayfish to frogs. To close the loop, her team did a more thorough survey of 97 Colorado wetlands, swabbing the skins of more than 9,000 amphibians. They found that those from wetlands with Orconectes crayfish were twice as likely to be infected with Bd, and that the presence of crayfish predicted the levels of infection better than anything else.
And then, the smoking gun: McMahon placed uninfected tadpoles in the same tanks as infected crayfish, and found that 70 percent of them picked up the fungus.
It’s an unparalleled suite of experiments. And it offers the first conclusive proof that Bd can infect hosts other than amphibians, in a way that eventually loops back to its favoured victims. “It was extremely important to me to make sure we did a very thorough job because I was worried others might be a little sceptical,” says McMahon. “[We wanted to present] the most concrete story possible.”
Now what?
Why is the fungus so wide-ranging in its tastes? Because of keratin, the substance found in your nails, skin and hair. Bd consumes keratin and related proteins in amphibian skins, and the same proteins line the guts of many invertebrates, like crayfish. “Given the appropriate conditions, like the right moisture and acidity, it’s possible that Bd could infect other organisms that contain the appropriate food source,” says McMahon.
The study threw up one more surprise: Bd doesn’t need to infect crayfish in order to kill them. The first clue was that the fungus seems to damage a crayfish’s gills, even though it infects its guts. When McMahon placed crayfish in water that had once contained Bd, but had been filtered to remove every spore, the animals still became sick. Clearly, the fungus releases some sort of chemical that can kill in its absence. Maybe it’s one of Bd’s several protein-destroying enzymes? Maybe it’s something else? Either way, it’s not clear if these chemicals could also kill amphibians at a distance.
Identifying a reservoir species seems like a blow for the many scientists who are trying to save amphibians from the fungus. If this frog-killer can hide out in other hosts, it will be more difficult to eradicate. But McMahon is optimistic. “It’s possible that managing alternative hosts offers a new and potentially more effective approach to managing Bd,” she suggests. Since crayfish are traded nationally and internationally for food and bait, and are often accidentally released, they could be helping to spread the fungus from place to place. If that’s true, then managing the spread of crayfish might help reduce the spread of Bd.
To Vredenburg, understanding these issues is crucial not just for saving amphibians, but for safeguarding our own health. It would be foolish for us to ignore a pandemic of this scale, even if it does not affect us directly. “There’s nothing like it in recorded history,” he says. “It makes the bubonic plague, which killed around 30 percent of the European population, look like nothing. We’re talking about 100 percent of not just one species, but probably hundreds.”
“Yes, this is occurring in amphibians, but they’ve been around for hundreds of millions of years, and are more historically successful than we are,” he adds. “We desperately need to understand how this could happen in a vertebrate group. We need to understand the biology that could lead to this global pandemic even though it’s not in humans or the species we rely on.”
Reference: McMahon, Brannelly, Chatfield, Johnson, Joseph, McKenzie, Richards-Zawackib, Veneskya & Rohr. 2012. Chytrid fungus Batrachochytrium dendrobatidis has nonamphibian hosts and releases chemicals that cause pathology in the absence of infection. PNAS http://dx.doi.org/10.1073/pnas.1200592110
More on amphibians:
- More Wolverine frogs! Japanese species uses bony thumb-spikes to fight and hold onto mates
- No, wait, THIS is the world’s smallest frog
- The olm: the blind cave salamander that lives to 100
- Newt healing factors unaffected by age and injury
- Frogs debug themselves by absorbing tracking devices into their bladders
- Climate change responsible for decline of Costa Rican amphibians and reptiles
- Coin-sized frog becomes mite-y thanks to poisonous diet
Go Further
Animals
- This ‘saber-toothed’ salmon wasn’t quite what we thoughtThis ‘saber-toothed’ salmon wasn’t quite what we thought
- Why this rhino-zebra friendship makes perfect senseWhy this rhino-zebra friendship makes perfect sense
- When did bioluminescence evolve? It’s older than we thought.When did bioluminescence evolve? It’s older than we thought.
- Soy, skim … spider. Are any of these technically milk?Soy, skim … spider. Are any of these technically milk?
- This pristine piece of the Amazon shows nature’s resilienceThis pristine piece of the Amazon shows nature’s resilience
Environment
- This pristine piece of the Amazon shows nature’s resilienceThis pristine piece of the Amazon shows nature’s resilience
- Listen to 30 years of climate change transformed into haunting musicListen to 30 years of climate change transformed into haunting music
- This ancient society tried to stop El Niño—with child sacrificeThis ancient society tried to stop El Niño—with child sacrifice
- U.S. plans to clean its drinking water. What does that mean?U.S. plans to clean its drinking water. What does that mean?
History & Culture
- Meet the original members of the tortured poets departmentMeet the original members of the tortured poets department
- Séances at the White House? Why these first ladies turned to the occultSéances at the White House? Why these first ladies turned to the occult
- Gambling is everywhere now. When is that a problem?Gambling is everywhere now. When is that a problem?
- Beauty is pain—at least it was in 17th-century SpainBeauty is pain—at least it was in 17th-century Spain
- The real spies who inspired ‘The Ministry of Ungentlemanly Warfare’The real spies who inspired ‘The Ministry of Ungentlemanly Warfare’
Science
- Here's how astronomers found one of the rarest phenomenons in spaceHere's how astronomers found one of the rarest phenomenons in space
- Not an extrovert or introvert? There’s a word for that.Not an extrovert or introvert? There’s a word for that.
- NASA has a plan to clean up space junk—but is going green enough?NASA has a plan to clean up space junk—but is going green enough?
- Soy, skim … spider. Are any of these technically milk?Soy, skim … spider. Are any of these technically milk?
Travel
- What it's like to hike the Camino del Mayab in MexicoWhat it's like to hike the Camino del Mayab in Mexico
- Is this small English town Yorkshire's culinary capital?Is this small English town Yorkshire's culinary capital?
- This chef is taking Indian cuisine in a bold new directionThis chef is taking Indian cuisine in a bold new direction
- Follow in the footsteps of Robin Hood in Sherwood ForestFollow in the footsteps of Robin Hood in Sherwood Forest