The remora is so ridiculous that no one would try to make it up. The top of its head is a giant, flat suction cup. It uses the cup to lock onto the bodies of bigger animals, such as sharks, sea turtles, and whales. As the big animal swims for miles in search of a meal, the remora hangs on for the ride. When its host finds a victim, the remora detaches and feasts on the remains. It sometimes cleans its host’s body and mouth of parasites, and then clamps its head back on for another ride.
The remora’s ridiculousness makes it a fascinating evolutionary puzzle just waiting for the solving. Other species clamp themselves onto other animals–whale barnacles, for example, grow prongs from their shells that anchor them to whale skin–but among fish, remoras are exceptional. Their closest relatives include Mahi-Mahi and amberjacks, neither of which has anything on their head that even faintly resembles the remora’s sucker. Only after the ancestors of remoras and these ordinary fish split apart some 50 million years ago, the remoras evolved a remarkably new piece of anatomy.

When you look closely at the remora’s suction disk, its remarkableness only grows. It looks like a spiked Venetian blind. Pairs of slat-like bones called lamellae form a series of rows running down the length of its head, and muscles running from the remora’s skull to those bones pivot them, creating spaces between the rows.
That negative pressure pulls the remora towards its host’s body. Each lamella also has a comb-like set of pins that help make its clamp even more secure. The whole structure is surrounding by a loose fleshy lip, ensuring that no water slips in, keeping the seal tight.
As a result, remoras can create a vacuum that’s not just strong enough to attach them to an animal, but to stay attached as water rushes past them. They can even hold tight as their hosts try to scrape them off on rocks. But a remora can instantly release itself when it’s time to eat, with just a flick of its muscles.
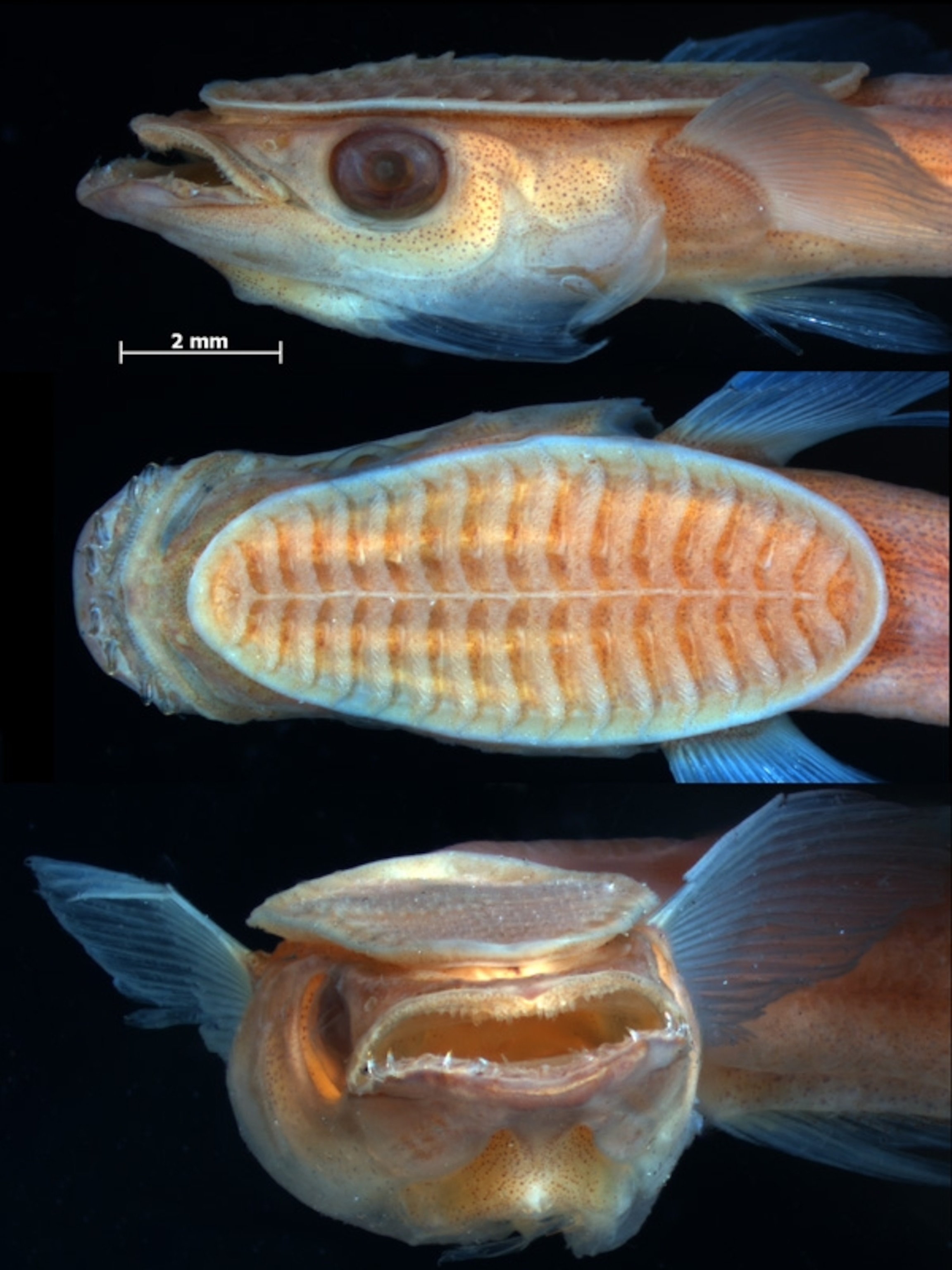
As impressive as the remora’s suction disk may be, however, it’s not actually all that new. As is so often the case in nature, it’s actually just evolutionary tinkering with old parts.
Scientists have gotten some clues to the remora’s origins by looking at how they grow up. When remoras hatch, they don’t yet have a suction disk. Last year Ralf Britz of the Natural History Museum in London and David Johnson of the Smithsonian made a careful study of young remoras to document their development. They found that the bones and muscles of the remora’s sucker start out much like the bones and muscles in a fin found on the back of other fish, known as the dorsal fin. They develop in the same location and have the same structure. But later, the bones and muscles move forward to the head.
They also change shape along the way. The fin spines spread out into lamellae that sprout a comb of spikes. In ordinary fish, the fin spine sits atop a small round bone. In remoras, that small bone widens out into another set of lamellae.
While the development of embryos doesn’t recapitulate evolution, it can offer some hints about how new things evolved from old ones. Britz and Johnson’s research indicates that the remora suction disk started out, improbably enough, as a dorsal fin. The fin stretched out into a complex vacuum device and moved up to the head. The underlying similarity between sucking disks and fins only becomes clear when you see how they both develop along the same path at first before diverging.
If all this were true, you might be able to test it by looking at the fossils of early relatives of remoras. Perhaps they captured the early stages of the transition.
That is precisely what Matt Friedman at the University of Oxford and his colleagues have done, as they report today in the Proceedings of the Royal Society.
Frieman is an expert on the larger group of fish to which the remora belongs, known as spiny rayed fishes. The group has evolved into some spectacularly weird forms, including sea horses and flatfishes. In 2008, I blogged about how Friedman found an intermediate flatfish, with one eye moving towards the other side. Remoras, with their own brand of weirdness, seemed to Friedman another spiny rayed fish worth spending some time on.
The only problem was that most of the fossils of remoras belonged to living lineages. Their suction disks were pretty much like what you’d find on a remora today. At least, that’s what most paleontologists who study remoras have thought.
Friedman decided to take a closer look at one of those remora fossils, called Opisthomyzon. The 30-million-year-old specimen was the first remora fossil ever found, in 1886, and it has sat in a museum in Switzerland ever since.

The specimen was in bad shape, Friedman found. It had a suction disk, for example, but it wasn’t clear if the whole disk had been pushed away from its original location. Friedman wondered if there might be other Opisthomyzon fossils hidden in other museums. Sometimes paleontologists can’t quite figure out what they’ve found, and they file away fossils without describing them.
At the National History Museum of London, Friedman found not one hidden Opisthomyzon fossil, but two. Mark Graham, a preparator at the museum, painstakingly chipped away at the underside of one of the new fossils, until all that was left was a paper-thin slab of rock. Friedman and his colleagues then compared the anatomy of Opisthomyzon to living remoras, as well as to extinct and living relatives, such as Mahi-Mahi.
Opisthomyzon proved to be exactly what Friedman was looking for: an extinct species that branched off before the origin of the living lineages of remoras. And when he and his colleagues examined its anatomy, they found exactly the kind of fish you’d expect to see from developing remoras: a fish with a suction disk still evolving from a fin.
This figure below sums up the story. The top fish is a conventional relative of remoras, with its dorsal fin bones shown to the right. In the middle is Opisthomyzon, with its corresponding suction bones. And at the bottom is a living remora.

You can see that the suction disk on Opisthomyzon is smaller than that of living remoras and does not sit over its whole head as it does today. The lamellae themselves bear more of a resemblance to the spines of dorsal fins. Opisthomyzon’s lamellae lacked a comb of spikes, for example, still retaining a single spine at the center.
Friedman’s research now gives us a richer hypothesis for how the remora got its sucker. Some of the remora’s closest living relatives, like cobia, tag along with bigger fish to scavenge on their scraps. The ancestors of remoras may have lived a similar life.
It’s not rare for spiny rayed fishes to grow extra dorsal fin spines. In the ancestors of remoras, such an anatomical fluke may have allowed them to latch their dorsal fin into the skin of a host fish, if only briefly. Even if they could spend a little time hitch-hiking this way, they would save energy that they’d otherwise have to spend on swimming for themselves.
Gradually, the remora’s dorsal fin became better adapted to latching onto other animals. As it moved towards the remora’s head, for example, it reduced drag. And as the fin bones spread outward, they attached the remora more strongly.
Sometimes, when we look at an adaptation in living animals, it seems to exquisitely well-suited to the animal’s life that we can’t imagine how a more primitive version of it could have provided any benefit. What good is half a wing, for example? What good is half a sucker? Fossils can give our limited powers of imagination a boost, by showing us that these intermediate forms did indeed exist. Opisthomyzon probably could ride on other animals, although it may have been more prone to get peeled off along the way. Remoras are so good at clamping onto their hosts that they’ve lost some of the traits that other fishes have. Their tails are weak, and they need a strong current of water passing over them in order to breathe through their gills. It’s probably no coincidence that Opisthomyzon had a much stronger tail than living remoras. It was only part-way down the road to ridiculousness.
Go Further
Animals
- Octopuses have a lot of secrets. Can you guess 8 of them?
- Animals
- Feature
Octopuses have a lot of secrets. Can you guess 8 of them? - This biologist and her rescue dog help protect bears in the AndesThis biologist and her rescue dog help protect bears in the Andes
- An octopus invited this writer into her tank—and her secret worldAn octopus invited this writer into her tank—and her secret world
- Peace-loving bonobos are more aggressive than we thoughtPeace-loving bonobos are more aggressive than we thought
Environment
- This ancient society tried to stop El Niño—with child sacrificeThis ancient society tried to stop El Niño—with child sacrifice
- U.S. plans to clean its drinking water. What does that mean?U.S. plans to clean its drinking water. What does that mean?
- Food systems: supporting the triangle of food security, Video Story
- Paid Content
Food systems: supporting the triangle of food security - Will we ever solve the mystery of the Mima mounds?Will we ever solve the mystery of the Mima mounds?
- Are synthetic diamonds really better for the planet?Are synthetic diamonds really better for the planet?
- This year's cherry blossom peak bloom was a warning signThis year's cherry blossom peak bloom was a warning sign
History & Culture
- Strange clues in a Maya temple reveal a fiery political dramaStrange clues in a Maya temple reveal a fiery political drama
- How technology is revealing secrets in these ancient scrollsHow technology is revealing secrets in these ancient scrolls
- Pilgrimages aren’t just spiritual anymore. They’re a workout.Pilgrimages aren’t just spiritual anymore. They’re a workout.
- This ancient society tried to stop El Niño—with child sacrificeThis ancient society tried to stop El Niño—with child sacrifice
- This ancient cure was just revived in a lab. Does it work?This ancient cure was just revived in a lab. Does it work?
- See how ancient Indigenous artists left their markSee how ancient Indigenous artists left their mark
Science
- Jupiter’s volcanic moon Io has been erupting for billions of yearsJupiter’s volcanic moon Io has been erupting for billions of years
- This 80-foot-long sea monster was the killer whale of its timeThis 80-foot-long sea monster was the killer whale of its time
- Every 80 years, this star appears in the sky—and it’s almost timeEvery 80 years, this star appears in the sky—and it’s almost time
- How do you create your own ‘Blue Zone’? Here are 6 tipsHow do you create your own ‘Blue Zone’? Here are 6 tips
- Why outdoor adventure is important for women as they ageWhy outdoor adventure is important for women as they age
Travel
- This royal city lies in the shadow of Kuala LumpurThis royal city lies in the shadow of Kuala Lumpur
- This author tells the story of crypto-trading Mongolian nomadsThis author tells the story of crypto-trading Mongolian nomads
- Slow-roasted meats and fluffy dumplings in the Czech capitalSlow-roasted meats and fluffy dumplings in the Czech capital
